|
Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol). 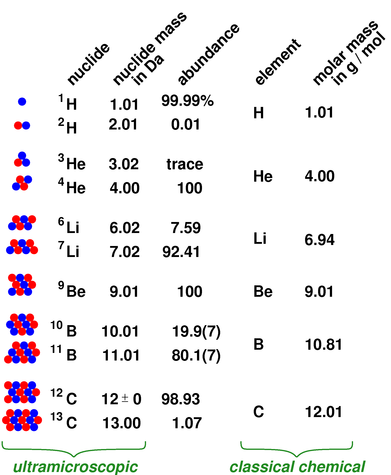
The molar mass has units of g/mol or kg/mol. A mole of substance is equal to Avogadros number (6.023×10 23) of that substance. However when talking about a mole of an ionic compound we will still use the term molar mass. Interactive periodic table with up-to-date element property data collected from authoritative sources. The molar mass is the mass of one mole of substance, whether the substance is an element or a compound. This is because there are no individual molecules in ionic compounds. Molar mass is not a detail included on the periodic table of elements because molar mass measures the mass of molecules and not atoms. For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. Become a member to unlock this answer Create your account. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. Design Copyright © 2017 Michael Dayah (). For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. The concept of molar mass can also be applied to compounds. The given mass of K (4. 
Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. It was first isolated in 1808 in England when Sir Humphry Davy electrolyzed a mixture of lime and. and is a reactive silvery metallic element found in Group 2 of the periodic table. This is approximately the sum of the number of protons and neutrons in the nucleus. By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams. Relative atomic mass The mass of an atom relative to that of carbon-12. We have defined a mole based on the isotopic atomic mass of carbon-12. Thermal Conductivity0.1805 W/ (m K) Specific Heat 14300 J/ (kg K) Heat Fusion 0.558 kJ/mol. The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. 
There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). Buy print or eBook Opens in a new window Book contents. This list contains the 118 elements of chemistry. Periodic table of the elements, with molar masses A Life Scientists Guide to Physical Chemistry. Plant Inspection & Process OptimalisationĬhemical elements listed by atomic mass The elemenents of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
